INTERCEPT® Blood System For Plasma
Pathogen Reduction System
Proven Pathogen Reduction with INTERCEPT Plasma
The INTERCEPT Blood System for Plasma delivers broad spectrum transfusion-transmitted infection (TTI) risk reduction by inactivating bacteria, viruses, parasites, and leukocytes.1 Plasma treated with the INTERCEPT Blood System Pathogen Reduction System (INTERCEPT Plasma), provides a proven safe and effective solution that helps protects patients,* improves plasma availability, and delivers value with operational benefits.
Help protect patients
INTERCEPT Plasma offers high levels of inactivation against a broad array of most clinically relevant pathogens, including established threats such as HIV-1, HBV, and HCV, along with certain emerging pathogens.1 INTERCEPT Plasma also inactivates leukocytes, providing an alternative to gamma irradiation for the prevention of transfusion-associated graft versus host disease (TA-GVHD).1


Bacteria
Gram-negative, Gram-positive, spirochaetes
- Klebsiella pneumoniae
- Enterobacter cloacae
- Pseudomonas aeruginosa
- Yersinia entreocolitica
- Staphylococcus epidermidis
- Staphylococcus aureus
- Treponema pallidum (Syphillis)
- Borrelia burgdoferi (Lyme disease)
- Anaplasma phagocytophilum (HGE agent)
Viruses
Gram-negative, Gram-positive, spirochaetes
- HIV-1 IIIB, cell-associated
- HIV-1 IIIB, cell-free
- DHBV (model virus for HBV)
- BVDV (model virus for HCV)
- HTLV-I
- HTLV-II
- West Nile virus (WNV)
- SARS-Associated Coronavirus
- Chikunguyna virus (CHIKV)
- Influenza A virus
- Parvovirus B19
- Bluetongue virus
- Adenovirus 5
Protozoan Parasites
- Plasmodium falciparum
- Babesia microti
- Trypanosoma cruzi
Leukocytes
- Human T-Cells
 Proven Safety and Efficacy
Proven Safety and Efficacy
The clinical performance, safety, and effectiveness of INTERCEPT Plasma have been demonstrated in multiple clinical trials and validated by international hemovigilance and routine. These studies showed consistent achievement of safety and efficacy endpoints, effective support for patients with complex transfusion needs, and no increase in adverse event rates during routine use.4-9
INTERCEPT Plasma not only provides robust protection against TTIs but also preserves key hemostatic properties essential for patient care. The retained activity of major coagulation factors confirms that therapeutic function is maintained, ensuring reliable plasma support for patients undergoing transfusion.1

 Improved Availability
Improved Availability
The availability of blood components continues to face a myriad of challenges, such as the threat of new, emerging pathogens. INTERCEPT Plasma helps maintain blood product continuity by remaining safe and accessible during potential outbreaks and pandemics.10

 Operational Benefits
Operational Benefits
INTERCEPT Plasma can be manufactured from both apheresis and whole blood derived plasma.

PROACTIVE APPROACH
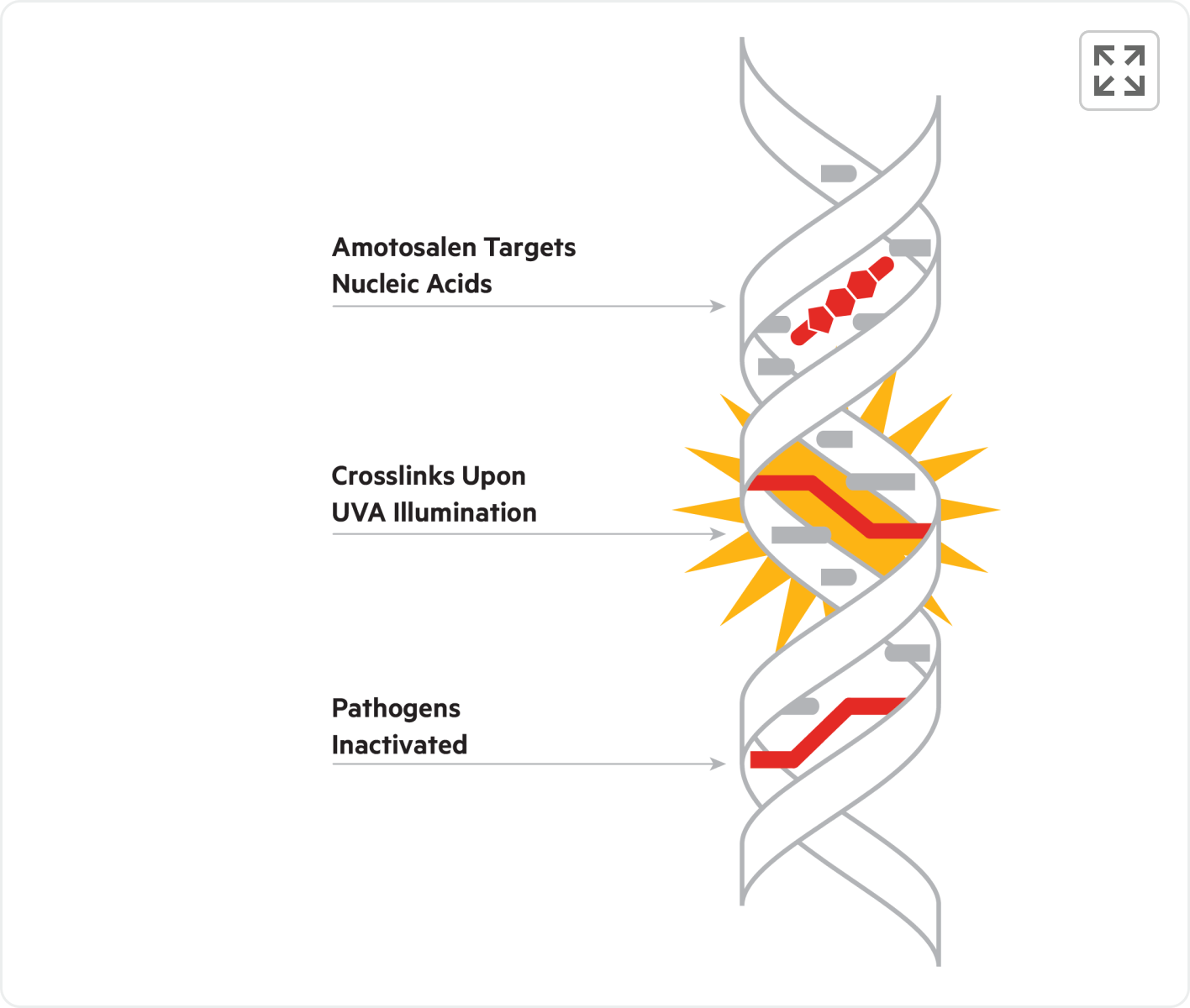
INTERCEPT Blood System Mechanism of Action
The INTERCEPT Blood System uses amotosalen, a well-characterized photoactive compound that targets DNA and RNA. With UVA illumination, amotosalen cross-links nucleic acids, blocking replication of pathogens and rendering them inactive.
You may also be interested in
*There is no pathogen inactivation process that has been shown to eliminate all pathogens. Certain non-enveloped viruses (e.g., HAV, HEV, B19, and poliovirus) and Bacillus cereus spores have demonstrated resistance to the INTERCEPT process. For a full list of pathogens, please refer to Package Insert.
References:
- The INTERCEPT Blood System for Plasma Package Insert, Cerus Corporation.
- Stramer SL, Hollinger FB et al. Transfusion 2009;49(S2):1S-29S.
- Dodd RY. Practical Transfusion Medicine. 4th ed. Chichester: Wiley; 2013;161-7.
- Hambleton J et al. Transfusion 2002;42:1302-1307.
- de Alarcon P et al. Transfusion 2005;45:1362-1372.
- Mintz PD et al. Blood 2006;107:3753-3760.
- Mintz PD et al. Transfusion 2006;46:1693-1704.
- Herbrecht R, et al. Characterization of efficacy and safety of pathogen inactivated and quarantine plasma in routine use for treatment of acquired immune thrombotic thrombocytopenic purpura. Vox Sang. 2018 May 21.
- Cazenave JP, et al. An active hemovigilance program characterizing the safety profile of 7483 transfusions with plasma components prepared with amotosalen and UVA photochemical treatment. Transfusion. 2010 Jun;50(6):1210-9.
- Allain, J.P., et al., Transfus Med Rev, 2005. 19(2): p. 110-26.