Pathogen Reduced Plasma, Cryoprecipitate Reduced
produced from the INTERCEPT® Blood System for Cryoprecipitation
Advancing Blood Safety and Innovation
Pathogen Reduced Plasma, Cryoprecipitate Reduced (PRPCR) is a pathogen reduced blood component for transfusion or therapeutic plasma exchange (TPE) in patients with thrombotic thrombocytopenic purpura (TTP).* Manufactured using plasma produced from the INTERCEPT Blood System for Plasma, PRPCR serves as a source of plasma proteins with reduced levels of fibrinogen, factor XIII, vWF, and factor VIII compared to unfractionated fresh frozen plasma (FFP), with a 5-day post-thaw shelf life.
Comprehensive Pathogen Reduction and Blood Safety
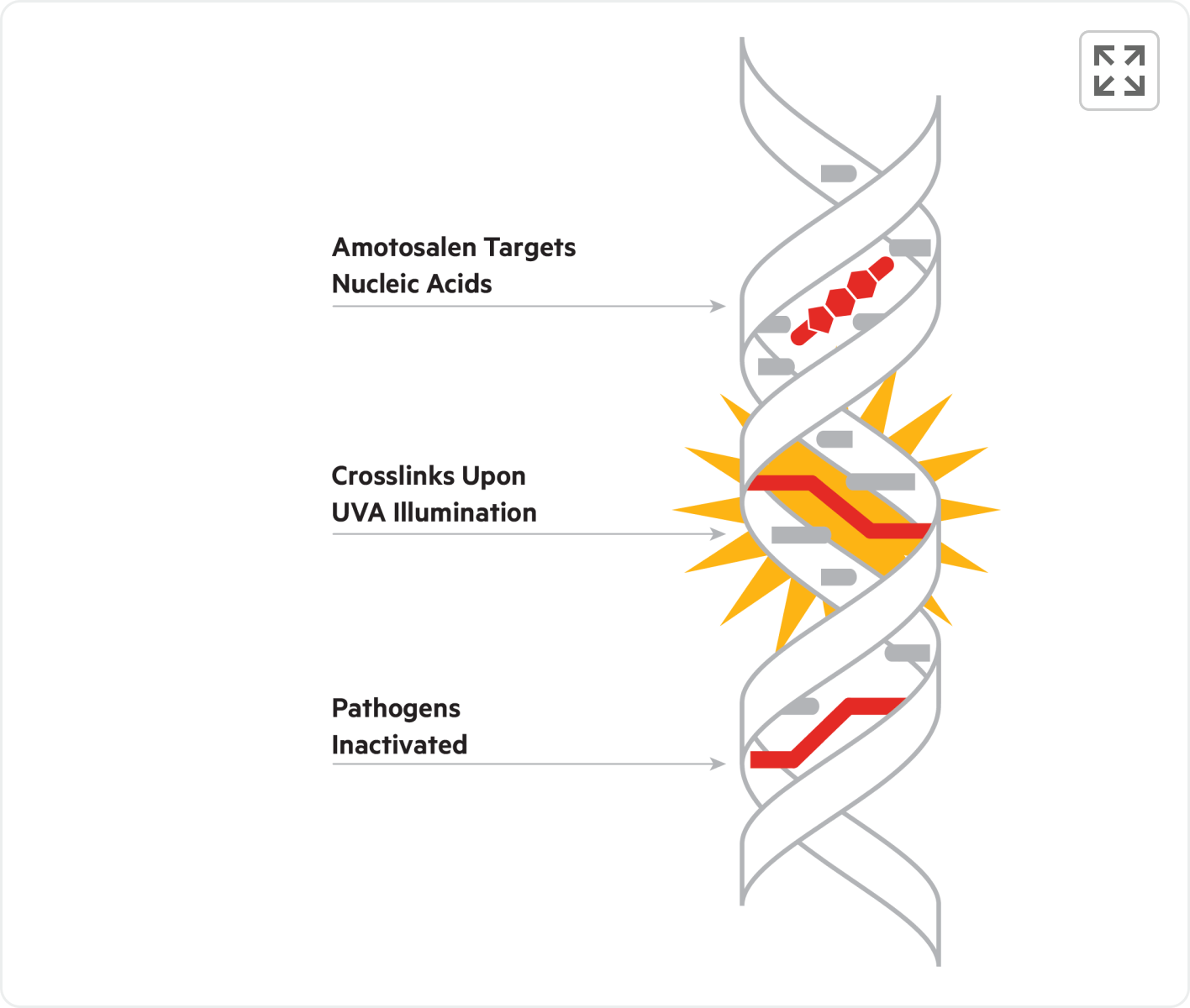
PRPCR is produced from pathogen reduced plasma treated with the INTERCEPT Blood System. As a result, PRPCR provides robust, broad spectrum transfusion-transmitted infection (TTI) risk reduction† by inactivating bacteria, viruses, parasites, and leukocytes.1,2
The INTERCEPT Blood System for Plasma uses amotosalen, a photoactive compound that targets nucleic acids. Upon exposure to UVA illumination, amotosalen forms irreversible cross-links between the nucleic acid strands, effectively preventing replication and rendering a broad spectrum of pathogens inactive.
This proactive approach to blood safety reduces infection risk to enhance clinical outcomes.
Availability and Operational Efficiency
PRPCR provides efficiency with its streamlined inventory and 5-day post-thaw room temperature shelf life.
Streamlined Inventory
Standardized single-use containers, provided as 240-295 mL units, simplify inventory management and east of use.
5-Day Post-Thaw Shelf Life
Store thawed product for up to five days at 1 to 6°C, with unused units easily returned to inventory and reallocated, minimizing waste.
12-Month Frozen Shelf Life
PRPCR arrives frozen with up to 12 months frozen shelf life.

5-Day Post-Thaw Shelf Life
You may also be interested in
*INTERCEPT Pathogen Reduced Plasma, Cryoprecipitate Reduced has not been evaluated in a clinical setting.
†There is no pathogen inactivation process that has been shown to eliminate all pathogens. Certain non-enveloped viruses (e.g., HAV, HEV, B19, and poliovirus) and Bacillus cereus spores have demonstrated resistance to the INTERCEPT process. For a full list of pathogens, please refer to Package Insert.
References: